PLANO, Texas - Oct. 26, 2021 - PRLog -- QinFlow, the manufacturer of the portable blood and IV warming Warrior product line, is proud to announce its ability to warm both blood and fluid when used in conjunction with the LifeFlow PLUS Blood & Fluid Infuser. The Warrior is a high-performance blood/IV fluid warmer for all pre-hospital and in-hospital emergency settings that features high performance, simplicity of operation, and portability, among others.
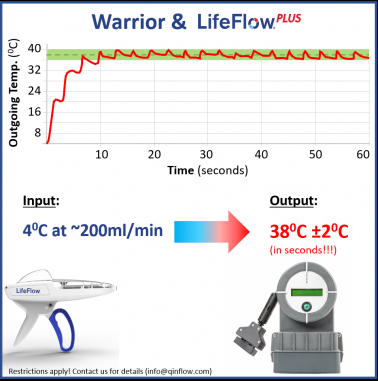
CEO Ariel Katz stated, "The Warrior is the only technology in the market that can effectively warm at rapid pulsatile flows due to a unique heat exchange process that provides excellent thermal transfer efficiency. The aluminum-free fluid path and highly robust mechanical structure provide additional layers of safety and durability. Our affordable consumable design provides commercial incentives and ensures best performance at the lowest cost."
He added, "We feel privileged to work together to provide an emergent resuscitation technology that enables emergency personnel to effectively treat the sickest patients when time is of the essence and minutes truly matter."
The LifeFlow PLUS is a portable, easy-to-use, device that allows faster and more efficient delivery of blood products and fluids when compared to traditional methods such as pressure bags or gravity infusion. Kyle Chenet, CEO of 410 Medical, commented, "An emergent trauma patient with hemorrhagic shock requires immediate blood transfusion. When used together LifeFlow PLUS and Warrior can deliver a unit of warmed blood products in two minutes, allowing the provider to quickly stabilize and reassess the patient."
About QinFlow
Since 2009 QinFlow (short for Quality in Flow) has worked to develop and perfect a proprietary fluid warming technology (patented) that delivers unparalleled levels of warming efficiency. The company's flagship line of products – the Warrior – provides front-end rescue teams, first responders, critical care transport teams, and emergency care professionals within the hospital with a high performance, reliable, simple to operate, and completely portable blood and IV fluid warming device that operates flawlessly in all environmental conditions in order to fight hypothermia and help in saving lives. QinFlow is headquartered in Rosh Ha'ayin (Israel) and Plano TX (USA). For more information on QinFlow and the Warrior modular system, visit www.qinflow.com. For more information regarding this press release and about our guidelines and disclaimers concerning the warming of intermittent flows, contact us at info@qinflow.com.
About 410 Medical
Based in Durham, NC, 410 Medical develops innovative products that enable frontline medical providers to improve care for critically ill patients. 410 Medical's first product family, the LifeFlow infusion system, is designed for patients in urgent need of rapid volume resuscitation. LifeFlow has been implemented at hospitals and EMS agencies across the United States. For more information visit www.410medical.com.
For more information on QinFlow and the Warrior line of warmers, visit www.qinflow.com or contact us at info@qinflow.com.
For more information on 410 Medical or on LifeFlow PLUS visit www.410medical.com or contact us at info@410medical.com.
Disclaimers
- This Press Release does not serve as a recommendation to use LifeFlow PLUS nor any other flow-inducing method. This decision remains at the sole discretion of the clinician.
- The Warrior configurations should be used in accordance with the applicable Instructions for Use (IFU) and labelling. In any case of conflict, the IFU guidelines shall govern. Users seeking to operate the devices in tandem with the LifeFlow PLUS rapid infuser should be familiar with each device/method use instructions, which should be followed at all times.
- Contact QinFlow and 410 Medical to learn about the guidelines and limitations of using each device.
Contact
Ariel Katz
***@qinflow.com
Photos: (Click photo to enlarge)

Read Full Story - QinFlow and 410 Medical Further Resuscitation Care through Warming and Rapid Blood and Fluid Delivery | More news from this source
Press release distribution by PRLog













