Theralase®'s Latest Preclinical Data Supports Combining Immunotherapy Drugs with Theralase® PDCs for Increased Cancer Destruction
TORONTO, ON / ACCESSWIRE / October 5, 2023 / Theralase® Technologies Inc. ("Theralase®" or the "Company") (TSXV:TLT)(OTCQB:TLTFF), a clinical stage pharmaceutical company dedicated to the research and development of Photo Dynamic Compounds ("PDCs"), in addition to the light and radiation systems that activate them, is focused on the safe and effective destruction of various cancers, bacteria and viruses, when light or radiation activated.
Theralase® announced that it has filed a US patent, titled "Enhanced Immunotherapeutic Method Comprising Combined Administration of Photodynamic / Radiotherapeutic Compounds and Immunotherapeutic Agents" for enhanced destruction of cancer.
The patent submission details combining Theralase®'s patented PDC technology with immunotherapy drugs for increased cancer kill and continues to build on Theralase®'s burgeoning intellectual property portfolio governing its PDCs, how they target cancer cells and how to best activate them safely and effectively to destroy cancer cells, while leaving healthy cells intact.
Over the last decade, immunotherapy drugs have delivered stellar results for some cancer patients; but, unfortunately zero benefit for others, with only 20 to 40% of patients deriving benefit, based primarily on the type of antigen that presents on an individual patient's cancer cells and the relatively low percentage (<25%) of patients, who display an effective anti-cancer immune response. For example, if the patient's cancer cells do not possess a specific antigen, then the immunotherapy drugs designed to bind to that antigen will have little to no benefit, with the patient's immune system remaining completely inactivated or suppressed, limiting efficacy for a majority of patients. In addition, immunotherapy drugs can be associated with serious or life-threatening side-effects.
Theralase®'s latest preclinical research supports the premise that both the safety and efficacy of immunotherapy drugs can be substantially increased in the destruction of cancer by allowing cancer cells to be more readily identified by the immune system.
The immune system defends and protects our bodies from infection and disease via specialized cells and chemical signal processes that work together to find and fight pathogens such as: viruses, bacteria, and abnormal cells that cause disease, such as cancer. Cancer cells have antigens on their surface that trigger the immune system to find and destroy them, but some antigens can allow them to hide from the immune system surveillance.
Examples of immunotherapy drugs, include: Chimeric Antigen Receptor ("CAR") T-cell therapy, cancer vaccines and immune checkpoint inhibitors, such as monoclonal antibodies.
It is therefore desirable to provide additional therapies to assist immunotherapy drugs to destroy these cancer cells for a whole-body approach in the destruction of cancer.
CD47 is the dominant macrophage checkpoint signal on cells that tell the immune system whether to destroy the cancer cells or leave them alone, thus CD47 is a powerful "Don't Eat Me" signal, overexpressed in many cancer cells that allows them to evade detection and hence destruction by Antigen Presenting Cells ("APCs"), such as macrophages and anti-cancer T cells. Researchers at Stanford have discovered that nearly every kind of cancer cell has a large quantity of CD47 expressed on their cell surface.
Theralase®'s latest preclinical research provides insight into how radiation activated RuvidarTM (TLD-1433) and Rutherrin® (TLD-1433 + transferrin) are able to significantly reduce the amount of CD47 that cancer cells express on their cell surface (See Figure 1), allowing the immune system to identify them and target them for destruction. This same process may allow immunotherapy drugs to increase their safety and efficacy in destroying cancer cells that have recently been "unmasked".
In other words, Theralase®'s PDCs are making these cancer cells much more visible to the immune system and thus much more susceptible to destruction both by Theralase®'s PDCs and by immunotherapy; hence, the ability to use combinational therapy.
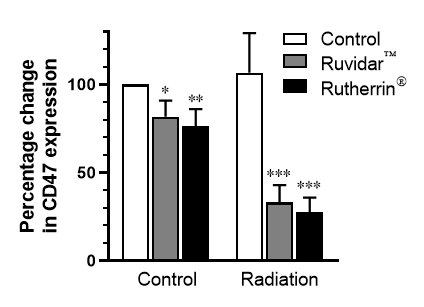
Note: The number of asterisks in Figure 1.0 denotes the statistical probability value (p value), with "***" indicating p<0.001 or a 0.1% that the observed difference could have occurred by chance alone.
In further developments of Theralase®'s preclinical research, increased extracellular Adenosine Tri Phosphate ("ATP") released by cells that have experienced Immunogenic Cell Death ("ICD") notifies the immune system to "Eat Me", which is an elegant way to amplify the killing of cancer cells.Theralase ®'s preclinical data suggests that both RuvidarTM and Rutherrin® are acting as strong ATP and ICD amplifiers for the immune system (See Figure 2).
Theralase®'s PDCs, as single agents, have been clinically proven to destroy cancerous tumours, when light activated and proven preclinically to activate an immune system response; hence, the combination of immunotherapy drugs with Theralase®'s PDC-based therapy introduces new opportunities for higher cancer cell kill, both locally and against distant tumours.
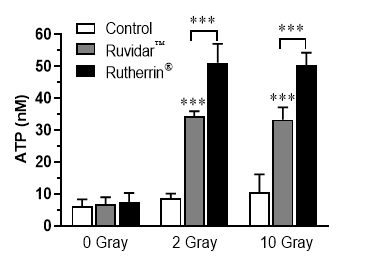
Dr. Arkady Mandel, MD, PhD, DSc, Chief Scientific Officer of Theralase® stated, "Theralase®'s latest preclinical research has demonstrated that our patented PDCs, RuvidarTM and Rutherrin®, are enabling targeted drug delivery and destruction of cancer in situ (in a localized tumour or anatomical state), but also activating local and systemic anti-cancer immune responses, for effective ongoing tumour destruction and increased anti-cancer resistance. CD47 plays a vital role in cancer cell proliferation, invasion and metastases, as it was found to be a key regulator in immunosurveillance and immune evasion in a large variety of cancers, including urinary bladder cancer. Reduction of CD47 expression by PDCs, such as RuvidarTM and Rutherrin® is one of the key targets of our research. Theralase®'s anti-CD47 therapy could sensitise tumours, such as urothelial carcinoma, to immunotherapy drugs. The results of our research may provide a potential strategy for direct tumour treatment by simultaneously targeting CD47 and PD-L1 checkpoints. The RuvidarTM and Rutherrin ®mediated amplification of ICD, ATP, "Find-Me" and "Eat-Me" signals, as well as suppression of CD47 checkpoint "Don't Eat Me" signal opens a new window for enhancing the effects of contemporary anti-cancer therapy. Based on the mechanism of action of Theralase®'s PDCs, strong safety profile and strong efficacy, as a single anti-cancer agent in our registered clinical study and the benefits of Rutherrin®, as an intravenous administered drug, particularly in combination with radiotherapy, in our preclinical research, it strongly supports achieving even higher tumour destruction and a complete response in patients by combining Theralase® PDCs with immunotherapy for various cancers."
Mr. Roger DuMoulin-White, BSc, P.Eng, Pro.Dir, President and Chief Executive Officer of Theralase® stated, "Theralase® has commenced the commercialization phase of the Company moving from the research and clinical development of its Phase II registration clinical study for bladder cancer to commercialization, distribution and partnering opportunities; however, Theralase® continues to strengthen its IP portfolio, through preclinical research, with additional patent submissions that support its anti-cancer therapy pipeline. We remain confident in achieving our strategic clinical and corporate milestones by year-end and into 2024 to create value for our shareholders. In the fourth quarter of 2023, Theralase® looks forward to presenting the latest clinical data from the Phase II Non-Muscle Invasive Bladder Cancer ("NMIBC") clinical study."
About Theralase® Technologies Inc.
Theralase®, a clinical stage pharmaceutical company dedicated to the research and development of Photo Dynamic Compounds ("PDCs"), in addition to the light and radiation systems that activate them, is focused on the safe and effective destruction of various cancers, bacteria and viruses, when light or radiation activated.
Additional information is available at www.theralase.com and www.sedar.com
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward Looking Statements
This news release contains Forward-Looking Statements ("FLS") within the meaning of applicable Canadian securities laws. Such statements include, but are not limited to, statements regarding the Company's proposed development plans with respect to Photo Dynamic Compounds ("PDCs") and their drug formulations. FLS may be identified by the use of the words "may, "should", "will", "anticipates", "believes", "plans", "expects", "estimate", "potential for" and similar expressions; including, statements related to the current expectations of the Company's management for future research, development and commercialization of the Company's PDCs and their drug formulations; including: preclinical research, clinical studies and development and regulatory approvals.
These statements involve significant risks, uncertainties and assumptions; including, whether the Company is able to: adequately fund and secure the requisite regulatory approvals to successfully complete preclinical and clinical studies in a timely fashion to implement its development plan; successfully commercialize its drug formulations; access sufficient capital to fund the Company's operations, which may not be available on terms that are commercially favorable to the Company or at all; provide preclinical and clinical support that the Company's drug formulations are effective against the conditions tested in its preclinical and clinical studies; comply with the term of license agreements with third parties, not to lose the right to use key intellectual property in its business; protect its intellectual property and the timing and success of this intellectual property and achieve acceptance and approval of regulatory filings. Many of these factors that will determine actual results are beyond the Company's ability to control or predict.
Readers should not unduly rely on these FLS, which are not a guarantee of future performance. There can be no assurance that FLS will successfully come to fruition, and as such, FLS involve known and unknown risks, uncertainties and other factors which may cause actual results or future events to differ materially from the FLS.
Although the FLS contained in the press release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these FLS.
All FLS are made as of the date hereof and are subject to change. Except as required by law, the Company assumes no obligation to update such statements.
For More Information:
1.866.THE.LASE (843.5273)
416.699.LASE (5273)
www.theralase.com
Kristina Hachey, CPA
Chief Financial Officer
khachey@theralase.com
416.699.LASE (5273) x 224
SOURCE: Theralase Technologies Inc.
View source version on accesswire.com:
https://www.accesswire.com/790186/theralaser-files-us-patent-for-enhanced-immunotherapy













