TORONTO, ON / ACCESSWIRE / June 19, 2024 / Theralase® Technologies Inc. ("Theralase®" or the "Company") (TSXV:TLT)(OTCQB:TLTFF), a clinical stage pharmaceutical company dedicated to the research and development of light and/or radiation activated small molecules and their formulations, intended for the safe and effective destruction of various cancers, bacteria and viruses, is pleased to announce that it's lead drug formulation, Rutherrin®, has demonstrated an ability to provide a complete response in a Non-Small Cell Lung Cancer ("NSCLC") animal model.
Theralase® recently press released its latest research, using a well-established Lewis Lung Cancer ("LLC1") orthotopic model, representing NSCLC. In this model, mouse lungs are implanted with lung cancer cells, inducing these mice to develop very aggressive, fast growing and metastatic lung tumors.
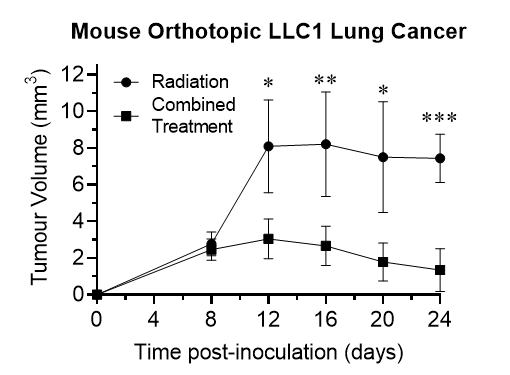
The mice were treated with x-ray radiation only as a control and x-ray activated Rutherrin® as the active arm. The mice treated with x-ray activated Rutherrin® demonstrated up to a 4-fold reduced tumor progression, based on Computerized Tomography ("CT") scan assessment of tumor volumes.
As shown in Figure 1, there was a significant delay in tumor progression in mice treated with x-ray activated Rutherrin® versus radiation (x-ray) alone (p> 0.001). In fact, in mice treated with x-ray activated Rutherrin®, the tumour is notably regressing / being destroyed over time.
As an update to this research, as shown in Figure 2, the Kaplan-Meier curve representing animal survival demonstrates a significant increase in overall survival of mice treated with x-ray activated Rutherrin® versus radiation only.
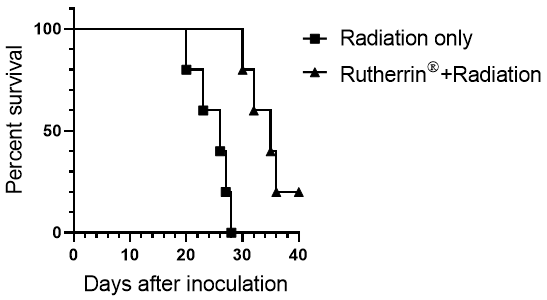
These results demonstrate that animals treated with a combination of Rutherrin® and radiation therapy showed an increase in median survival from 26 to 35 days, versus radiation only. In scientific publications, mouse survival of 9 days has been equated to the equivalent of 1 year survival in humans, but more importantly, is that one animal treated with the x-ray activated Rutherrin® (which had a positive lung tumor verified by CT scan) demonstrated a complete response and is now considered cancer free.
Dr. Arkady Mandel, M.D., Ph.D., D.Sc., Chief Scientific Officer of Theralase® stated, "The latest preclinical research demonstrates the ability of x-ray activated Rutherrin® to eradicate lung cancer in an animal model. This is initial research and through optimization, Theralase® hopes to achieve a complete response in a much greater percentage of animals, but this initial data is extremely encouraging. As a result of this latest success, Theralase® has committed to bringing this technology to market through the systematic research and development of this cutting-edge clinical therapy to safely and effectively destroy various cancers in patients. As a direct result, our list of cancer targets has increased from bladder cancer, which in the late stage of clinical development, to encompass brain cancer, lung cancer and various blood-based cancers, such as: leukemia, lymphoma and multiple myeloma."
Roger DuMoulin-White, B.E.Sc., P.Eng., Pro.Dir., President and Chief Executive Officer of Theralase® stated, "This latest research symbolizes the significant progress Theralase® has made over the last few years in the research and development of world-class systemic and targeted therapies for the treatment of various hard-to- treat cancers, such as: bladder cancer, brain and lung cancer. Pending sufficient capitalization and completion of a Good Laboratory Practice ("GLP") toxicology analysis for Rutherrin®, Theralase® plans to commence clinical studies for brain cancer, lung cancer and various blood-based cancers. If proven safe and effective in humans, Theralase® hopes to change the paradigm of how patients diagnosed with cancer are treated in the future."
About Lung Cancer:
Lung cancer is the leading cause of cancer death worldwide. Most patients die of progressive metastatic disease despite aggressive local and systemic therapies. The survival rate for lung cancer depends on the type, stage and age of the patient, with the overall 5-year survival rate for all types of lung cancer about 26.6%. Lung cancer is histologically classified into two main types: Small Cell Lung Cancer ("SCLC"), which accounts for approximately 15% of the patients diagnosed with lung cancer and Non-Small Cell Lung Cancer ("NSCLC"), which accounts for approximately 85%.
About Theralase® Technologies Inc.:
Theralase® is a clinical stage pharmaceutical company dedicated to the research and development of light and/or radiation activated small molecule compounds, their associated drug formulations and the light systems that activate them, with a primary objective of efficacy and a secondary objective of safety in the destruction of various cancers, bacteria and viruses.
Additional information is available at www.theralase.com and www.sedar.com
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward Looking Statements:
This news release contains "forward-looking statements" within the meaning of applicable Canadian securities laws. Such statements include; but, are not limited to statements regarding the Company's proposed development plans with respect to small molecules and their drug formulations. Forward looking statements may be identified by the use of the words "may, "should", "will", "anticipates", "believes", "plans", "expects", "estimate", "potential for" and similar expressions; including, statements related to the current expectations of Company's management for future research, development and commercialization of the Company's small molecules and their drug formulations, preclinical research, clinical studies and regulatory approvals.
These statements involve significant risks, uncertainties and assumptions; including, the ability of the Company to fund and secure the regulatory approvals to successfully complete various clinical studies in a timely fashion and implement its development plans. Other risks include: the ability of the Company to successfully commercialize its small molecule and drug formulations, the risk that access to sufficient capital to fund the Company's operations may not be available on terms that are commercially favorable to the Company or at all, the risk that the Company's small molecule and drug formulations may not be effective against the diseases tested in its clinical studies, the risk that the Company's fails to comply with the terms of license agreements with third parties and as a result loses the right to use key intellectual property in its business, the Company's ability to protect its intellectual property, the timing and success of submission, acceptance and approval of regulatory filings. Many of these factors that will determine actual results are beyond the Company's ability to control or predict.
Readers should not unduly rely on these forward-looking statements, which are not a guarantee of future performance. There can be no assurance that forward-looking statements will prove to be accurate as such forward-looking statements involve known and unknown risks, uncertainties and other factors which may cause actual results or future events to differ materially from the forward-looking statements.
Although the forward-looking statements contained in the press release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these forward-looking statements.
All forward-looking statements are made as of the date hereof and are subject to change. Except as required by law, the Company assumes no obligation to update such statements.
For investor information on the Company, please feel to reach out Investor Inquiries - Theralase Technologies.
For More Information:
1.866.THE.LASE (843-5273)
416.699.LASE (5273)
www.theralase.com
Kristina Hachey, CPA
Chief Financial Officer
X 224
khachey@theralase.com
SOURCE: Theralase Technologies Inc.
View the original press release on accesswire.com













